 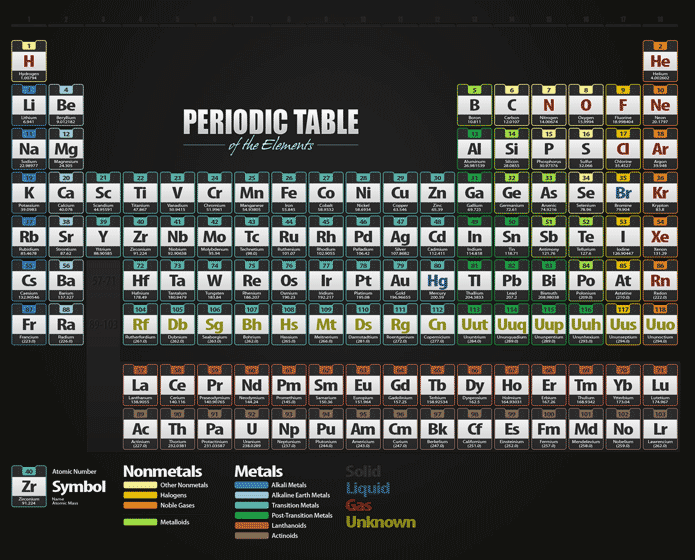
This shows how important it is to understand the fundamentals of atomic models and energy levels. Cambridge University Press has reported that more than 97% of the known matter in the universe is made up of atoms. 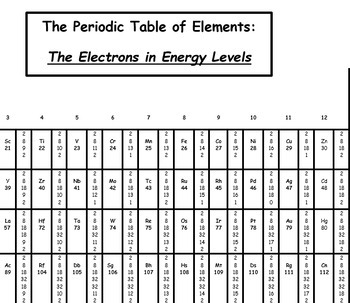
Industry Statistics That Support the Importance of This Topic This information is also important for fields such as chemistry, materials science, and even electronics engineering. Understanding energy levels and electron organization helps explain the chemical properties of different elements and can be used to predict how atoms will react. Why is This Information Important?Įmpower your business by understanding commercial electricity rates with our comprehensive guide.Knowing how many electrons can the first energy level hold is important for understanding the basics of atomic models and how atoms interact with each other. Electrons must fill energy levels in a specific order, starting with the first energy level and moving outward.The first energy level can hold up to two electrons.The first energy level is closest to the nucleus and has the lowest energy.The number of electrons an energy level can hold depends on the energy of that level.This allows for a predictable way for electrons to fill up an atom and helps explain why elements in the same group on the periodic table have similar chemical properties. The same is true for electrons and energy levels you can't move to the second energy level until the first is filled. You can't go to the second step until you've first stepped on the first step. Unlock the secrets of affordable energy in the Lone Star state with our guide on how to find the best cheap electricity providers in Texas.Imagine you have a staircase with five steps. Electrons must fill energy levels in a specific order, and the first energy level is the first step in that process. The reason the first energy level can only hold two electrons is because of the way electrons are organized within an atom. This might seem like a small number, but it's important for understanding how atoms interact with each other. Now that we know the basics of atomic models and energy levels, let's answer the question at hand: how many electrons can the first energy level hold? The answer is simple: the first energy level can hold up to two electrons. How Many Electrons Can the First Energy Level Hold? 
It's followed by the second energy level, which has slightly higher energy, and so on. The first energy level is closest to the nucleus and has the lowest energy. The energy of each level increases as it gets farther from the nucleus. Each model has its way of describing the location of electrons and the size and shape of the atom.ĭiscover how to effortlessly find affordable energy providers by checking out these 5 easy ways to find your next cheap electricity supplier.Energy levels are specific regions surrounding the nucleus where electrons can be found. There are several atomic models, including the Bohr model, the quantum mechanical model, and the electron cloud model. But how many electrons can the first energy level hold, and why is it important to know this information? Let's dive into the world of atomic models and find out! The Basics of Atomic Models and Energy Levelsīefore we get to the first energy level, it's important to understand how atomic models and energy levels work. The electrons are organized into energy levels, and each level can hold a specific number of electrons. Each such orbital can be occupied by a maximum of two electrons, each with its own projection of spin m s. As an alternative to the magnetic quantum number, the orbitals are often labeled by the associated harmonic polynomials (e.g., xy, x 2 − y 2). Įach orbital in an atom is characterized by a set of values of the three quantum numbers n, ℓ, and m l, which respectively correspond to the electron's energy, its angular momentum, and an angular momentum vector component ( magnetic quantum number). The term atomic orbital may also refer to the physical region or space where the electron can be calculated to be present, as predicted by the particular mathematical form of the orbital. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. In atomic theory and quantum mechanics, an atomic orbital ( / ˈ ɔːr b ɪ t ə l/) is a function describing the location and wave-like behavior of an electron in an atom. To see the elongated shape of ψ( x, y, z) 2 functions that show probability density more directly, see pictures of d-orbitals below. Each picture is domain coloring of a ψ( x, y, z) function which depends on the coordinates of one electron. The two colors show the phase or sign of the wave function in each region. 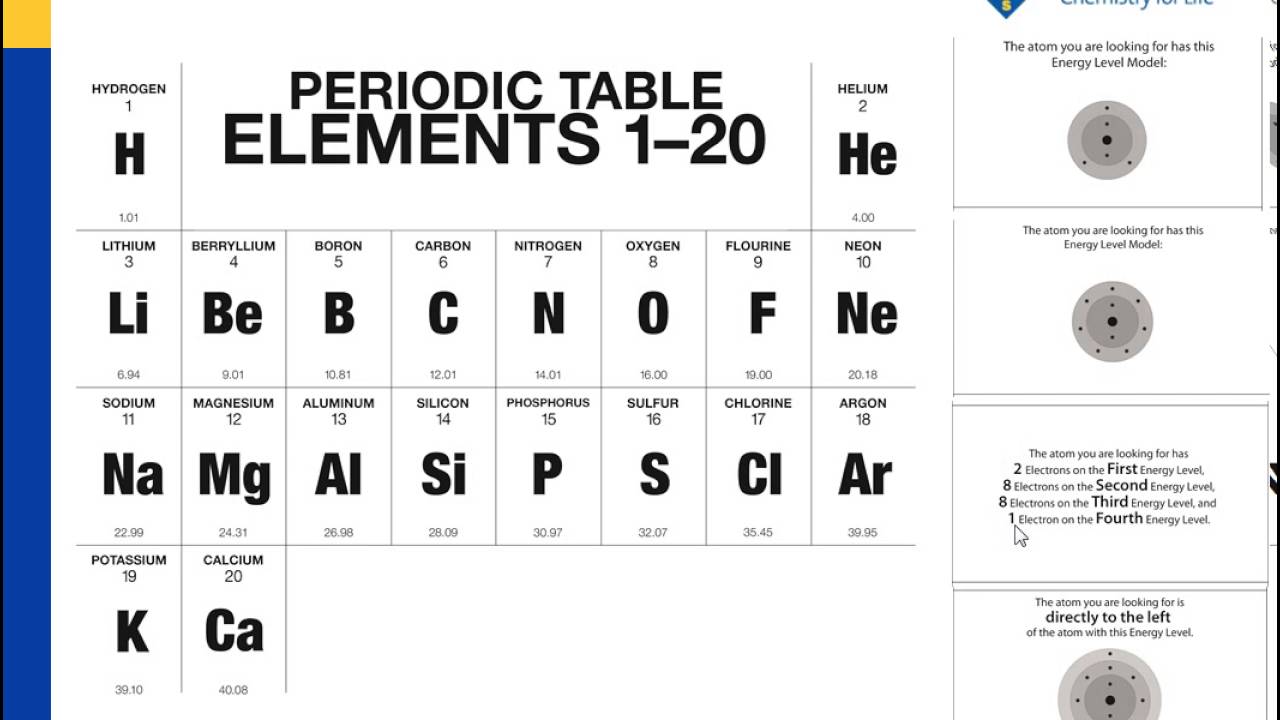
The shapes of the first five atomic orbitals are: 1s, 2s, 2p x, 2p y, and 2p z. For the collection of spaceflight orbits, see Orbital shell (spaceflight). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
